So What Is an Exosome, Exactly?
An exosome is a tiny, membrane-wrapped vesicle essentially a nano-sized package that cells release as a form of communication. Every cell in your body produces them. They're not waste products (though that was an early theory). They're more like biological text messages: packages of proteins, lipids, and genetic material (like microRNAs) sent from one cell to another to coordinate behavior.
A 2024 review in Signal Transduction and Targeted Therapy one of the highest-impact journals in the field, published under Nature describes exosomes as possessing key advantages over whole stem cell therapies, including non-immunogenicity, non-infusion toxicity, easy access, and freedom from tumorigenic potential. The authors note that exosomes can inherit therapeutic effects from their parent cells through the delivery of their signaling cargo.[A1]
A 2025 review in Pharmaceutics the first of its kind to systematically compare exosomes by cellular source describes them as carrying a variety of bioactive molecules that can be delivered to target cells, noting low immunogenicity and high biosafety as properties driving their application potential in tissue engineering, wound healing, and disease intervention.[A2]
Think of exosomes as your cells' messaging system except instead of texts, they're sending proteins and genetic instructions, in packages smaller than most viruses.
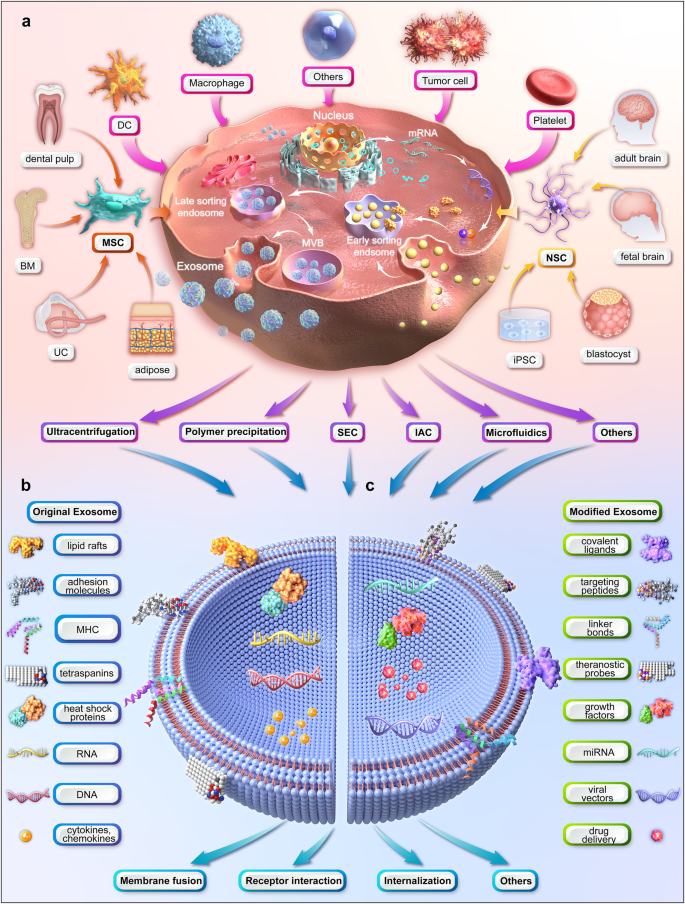
Where Do They Come From? Source Matters More Than You'd Think
Not all exosomes are created equal and this is one of the most important things to understand when evaluating any exosome-based application. Exosomes take on characteristics of the cell that produces them. An exosome from a mesenchymal stem cell (MSC) carries a very different cargo than one from a skin cell, a platelet, or a neural cell.
The 2025 Pharmaceutics review explicitly introduces a cell type-oriented framework for selecting exosome producers, calling it the first comprehensive comparison of exosomes based on cellular source and noting that factors like donor site and collection method also influence the therapeutic cargo.[A2]
For regenerative applications, MSC-derived exosomes have drawn the most research attention. A 2024 review in Clinical and Experimental Medicine specifically examines the research landscape around MSC-derived exosomes, describing active investigation into their signaling properties across inflammation, cellular communication, and tissue biology with MSCs from bone marrow, adipose tissue, and umbilical cord tissue being the most studied sources. The authors note these findings are preliminary and not yet constitutive of approved clinical indications.[A3]
As of January 2025, a review in Signal Transduction and Targeted Therapy notes that MSCs are the most widely used source for EV-based clinical trials globally accounting for roughly 61% of interventional EV studies registered on ClinicalTrials.gov.
The Aesthetic Medicine Connection
Aesthetic medicine the clinical field concerned with skin health, tissue quality, hair restoration, and non-surgical rejuvenation has become one of the most active areas of exosome research interest. The reason is fairly intuitive: skin and connective tissue are highly dynamic biological environments where cellular communication plays a central role in repair, renewal, and aging.
In aesthetic and dermatological contexts specifically, the research interest centers on exosomes' potential to influence collagen synthesis pathways, support wound healing, modulate inflammatory responses in the skin, and interact with fibroblasts and keratinocytes the cell types most relevant to skin structure and texture.
"Exosomes don't act on skin the way a topical cream does. They work at the cellular communication level influencing how cells behave and what proteins they produce."
A comprehensive 2025 analysis in the Journal of Pharmacology and Pharmacotherapeutics examined 90 registered human clinical trials involving exosomes. The analysis found that therapeutic roles represented approximately 21% of trials, with MSC-derived exosomes prominently featured for applications involving tissue repair and cellular signaling. The authors note steady clinical output from 2020 to 2024 with rising interest across specialties.[A4]
MSC-Derived Exosomes and Age-Related Cellular Changes
A 2025 review in Cell Proliferation (Wiley) specifically examines MSC-derived exosomes in the context of age-related diseases. The review discusses the role of MSC-derived exosomes in modulating cellular senescence pathways the processes by which cells age and decline in function and their involvement in tissue homeostasis. The authors highlight exosomal microRNA and protein cargo as key mechanisms through which these effects may be mediated.[A5]
Cellular senescence is relevant to aesthetic medicine because it underlies many of the visible signs of skin aging loss of elasticity, thinning, altered texture. Research into whether exosomes can influence senescence pathways in skin cells is an active and scientifically legitimate area of inquiry, though clinical translation is still in early and intermediate stages.
What the Clinical Trial Landscape Looks Like Right Now
As of early 2025, there are 292 EV-related clinical trials registered on ClinicalTrials.gov, including 170 interventional studies. Of those, 117 are specifically designed to assess therapeutic effects across disease areas including skin, inflammatory, and nervous system conditions. The FDA approved its first investigational new drug application for an EV-based candidate (AB126, from Aruna Bio) in January 2024 a milestone for the field. Korea's Ministry of Food and Drug Safety approved a Phase 1b trial for an umbilical cord MSC-derived EV product in May 2025.
This activity tells us that exosome science is being taken seriously by major regulatory bodies, academic medical centers, and pharmaceutical developers not just wellness companies. It also tells us we're in an early-to-mid stage of clinical development, not at the finish line.
90+ human trials. 292 registered EV studies globally. The FDA's first IND approval for an exosome-based candidate. The science is real and it's moving.
What the Science Does and Doesn't Say
It's worth being honest about where the science currently stands, because the gap between basic research and clinical proof-of-concept is where a lot of misinformation lives.
Most exosome research in aesthetic contexts is at the preclinical stage (cell cultures and animal models) or early Phase I/II human trial stage. Rigorous, large-scale, placebo-controlled human trials with standardized exosome preparations are still limited. The field is also grappling with meaningful technical challenges: there is currently no fully standardized method of exosome isolation, characterization, or potency testing which means the quality and consistency of exosome preparations can vary significantly depending on source, production method, and facility.
The 2025 Pharmaceutics review explicitly notes that clinical application requires selecting cellular sources based on specific intent while optimizing influencing factors suggesting there is no universal exosome preparation appropriate for all uses.[A2]
Regulatory status also varies: in the United States, exosome products used in humans are generally regulated by the FDA as biologics, and the rules around their clinical use, marketing claims, and commercialization are actively evolving.
Smart Questions to Ask Your Practitioner
If you're working with a practitioner who uses or recommends exosome-based products, these are scientifically grounded questions worth raising:
The Bottom Line
Exosomes are a legitimately exciting area of biomedical research. The science behind cellular communication, extracellular vesicle biology, and MSC-derived signaling is real, peer-reviewed, and growing rapidly. In aesthetic medicine specifically, the interest is scientifically justified but the clinical evidence base is still maturing, and the regulatory landscape is still evolving.
The most intellectually honest position is this: exosomes represent a promising frontier, with a credible scientific foundation and a growing clinical trial record. They're not magic, and they're not hype they're biology, which is to say, more interesting than either.
Educational Disclaimer: This article is intended for informational purposes only and does not constitute medical advice, diagnosis, or treatment recommendations. All information is drawn from peer-reviewed scientific literature. Exosome-based therapies are an active area of research; regulatory status and clinical availability vary by jurisdiction. Consult a licensed healthcare provider before making any medical decisions.
Tan F, et al. Clinical applications of stem cell-derived exosomes. Signal Transduction and Targeted Therapy. Jan 2024;9:17. DOI: 10.1038/s41392-023-01704-0.
View source →Chen Y, et al. Exosome Source Matters: A Comprehensive Review from the Perspective of Diverse Cellular Origins. Pharmaceutics. Feb 2025;17(2):147. PMCID: PMC11858990.
View source →Therapeutic potential of MSC-derived exosomes. Clinical and Experimental Medicine. Mar 2024. PMCID: PMC10907468. DOI: 10.1007/s10238-023-01282-z.
View source →Mohan S, et al. Clinical Frontiers of Exosome Research: A Comprehensive Analysis of Human Trials in Diagnostics, Therapeutics, and Regenerative Medicine. Journal of Pharmacology and Pharmacotherapeutics. 2025. DOI: 10.1177/0976500X251361201.
View source →MSC-Derived Exosomes for Age-Related Diseases. Cell Proliferation. May 2025. PMCID: PMC12099225.
View source →