WHY THIS MATTERS
The COA Is the Document That Separates Real Biologics From Marketing
Exosome products are showing up in med spas, dermatology offices, hair restoration clinics, and dermatologic clinical trials at a rate that exceeds the regulatory clarity of the category. Some of those products are genuinely well-characterized. Others arrive with a glossy brochure, a vague reference to "stem cell technology," and a one-page document that calls itself a Certificate of Analysis but reads more like a marketing tear-sheet.
For a licensed practitioner, the Certificate of Analysis is the single most important piece of paper in the supplier relationship. It is what allows a clinician to defend a treatment choice in a chart note, in a peer-review setting, in front of a state medical board, and in front of a patient who deserves to know what is being applied to them. Reading a COA the way a chemist reads it, by checking the assays, the units, the methods, and the negative controls, is a core practitioner competency in this category.
This article walks through the science: which surface markers a real exosome carries, which microRNAs are doing the biological work, which mesenchymal stem cell sources produce which cargo profiles, and which clinical trial endpoints are starting to translate the preclinical signal into measurable outcomes. Then it walks through the COA itself, parameter by parameter, with the analytical framework MISEV 2023 has codified for the field.
| 30 to 150 nm Exosome size range | ≥ 3 markers MISEV 2023 minimum (CD9, CD63, CD81) | < 5 EU/kg/hr Standard endotoxin limit, USP <85> |
THE BIOLOGY
What an Exosome Actually Is
An exosome is a small extracellular vesicle, between 30 and 150 nanometers in diameter, formed inside the parent cell through the endosomal pathway. Late endosomes mature into multivesicular bodies (MVBs), which contain intraluminal vesicles. When an MVB fuses with the plasma membrane, those intraluminal vesicles are released into the extracellular space as exosomes. This biogenesis pathway distinguishes them from microvesicles, which bud directly from the plasma membrane, and from apoptotic bodies, which are released during cell death [1].
The therapeutic interest in exosomes is inseparable from their cargo. Each vesicle carries a complex mixture of proteins, lipids, mRNAs, and microRNAs that reflects the biology of the parent cell. When an exosome reaches a recipient cell through fusion, receptor binding, or endocytosis, it delivers a multi-pathway biological message. A 2024 review in Signal Transduction and Targeted Therapy describes exosomes as inheriting the therapeutic effects of their parent cells through this delivery, while avoiding the risks associated with live cell administration [1].
For practitioners, three categories of biology deserve attention before reading any COA: the surface markers that confirm exosome identity, the microRNA cargo that drives the downstream biological effect, and the parent cell source that determines what the cargo looks like in the first place.
SURFACE MARKERS
The Tetraspanin Triad: CD9, CD63, and CD81
Three tetraspanins, CD9, CD63, and CD81, are the canonical surface markers used to identify exosomes. They belong to a four-transmembrane-domain protein family that organizes membrane microdomains called tetraspanin-enriched microdomains (TEMs). Each of the three carries different biological roles in the parent cell, occupies different subcellular compartments, and contributes differently to vesicle behavior [2].
Figure 1. The MISEV 2023 framework for exosome identification requires at least three positive markers and one negative marker. CD9, CD63, and CD81 are the canonical tetraspanin triad.
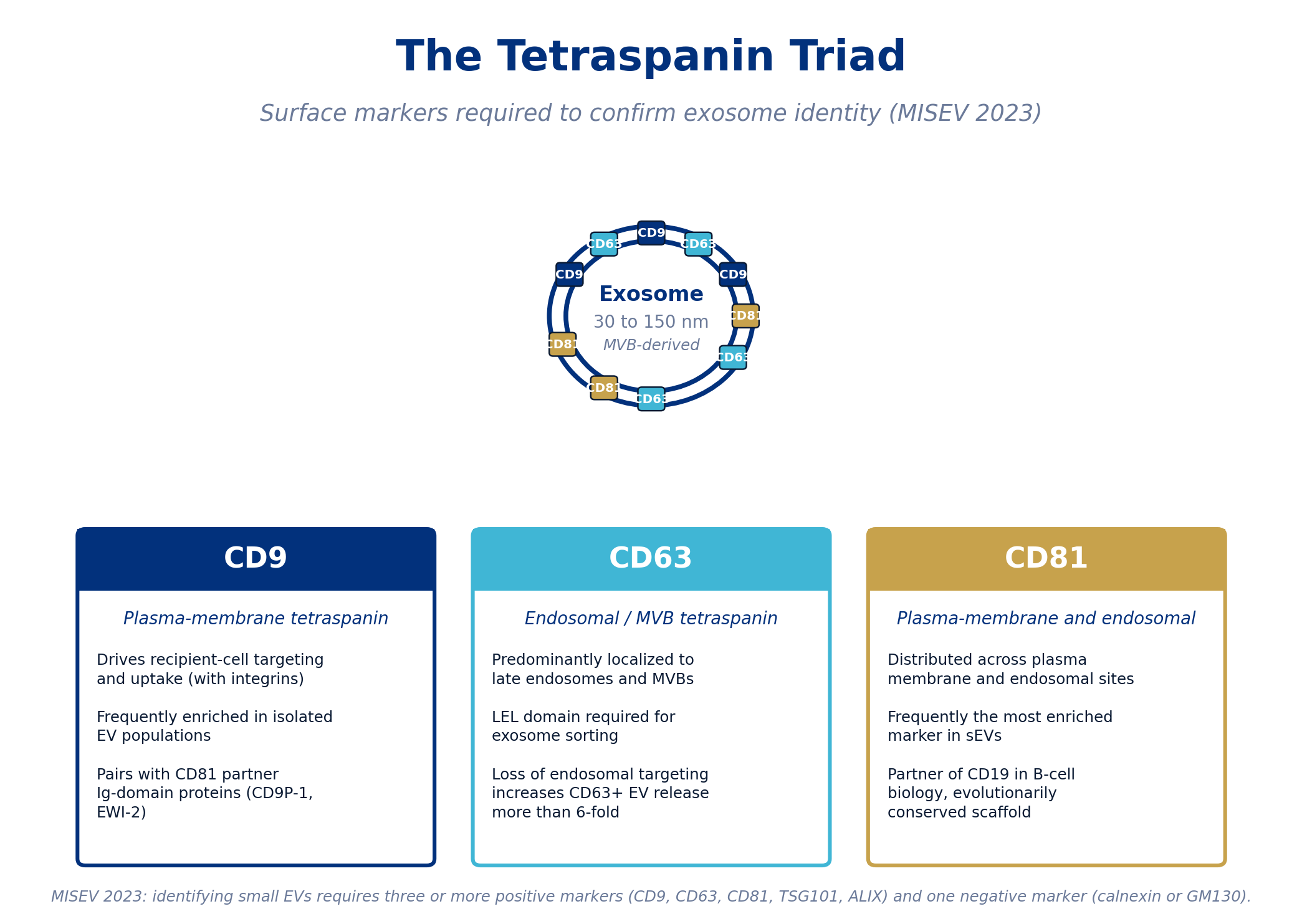
CD9: Plasma-Membrane Tetraspanin Driving Recipient-Cell Targeting
CD9 is predominantly localized to the plasma membrane of the producing cell, and it appears at the surface of secreted exosomes. Its biological role is closely tied to recipient-cell targeting and uptake. CD9, together with integrin αvβ3, has been shown to mediate the targeting of exosomes to dendritic cells [2]. CD9 frequently associates with partner Ig-domain proteins, including CD9P-1 and EWI-2, which co-segregate into EVs and may participate in cell adhesion and signaling [3]. Functionally, CD9-positive vesicles are enriched in many isolated EV preparations, and CD9 is one of the most reliably detected positive markers across MSC sources.
CD63: The Endosomal Marker That Defines Exosome Origin
CD63 is the tetraspanin most tightly associated with the endosomal compartment. In producing cells, CD63 is mainly confined to multivesicular bodies, which is exactly the compartment where exosomes form [2]. The large extracellular loop (LEL) of CD63 carries a critical sorting signal: when the LEL is deleted, CD63 fails to localize to exosomes [4]. Conversely, C-terminal modifications that disrupt endosomal targeting cause CD63 to relocalize to the plasma membrane and produce a more than six-fold increase in CD63-positive EV release [3]. Beyond the marker role, CD63 is involved in cargo sorting (a 2014 study identified CD63-dependent EV targeting in neuroblastoma cells [2]) and, through its complex with syntenin-1, in the trafficking of certain viral proteins [3].
CD81: The Most-Enriched Marker, With a Conserved Scaffolding Role
CD81 is distributed across both the plasma membrane and endosomal compartments, and it is frequently the most enriched of the three tetraspanins in isolated small EV preparations [3]. CD81 plays well-characterized roles outside the EV field, including its partnership with CD19 in B-cell costimulation. CD81-deficient mice and humans show impaired humoral immunity through decreased CD19 expression [3]. In the EV context, CD81 forms a scaffolding complex with the same Ig-domain proteins (EWI-F and EWI-2) that partner with CD9, and the loss of both CD9 and CD81 simultaneously causes a measurable depletion of these partners in EVs.
MISEV 2023 minimum: Identification of small extracellular vesicles requires at least three positive markers, drawn from the tetraspanins (CD9, CD63, CD81) plus optional candidates such as TSG101 or ALIX, together with at least one negative marker (calnexin, GM130, or another non-vesicular protein) to confirm low cellular debris contamination [5]. A COA that reports only one tetraspanin does not meet this minimum.
CARGO BIOLOGY
MicroRNA Cargo: The Working End of the Therapy
Surface markers confirm what an exosome is. MicroRNAs explain what an exosome does. A 2024 review of MSC-derived exosomes describes their miRNA cargo as a reproducible, cell-type-specific repertoire that delivers measurable downstream effects on gene expression once internalized by recipient cells. The miRNAs do not act independently; they cluster around a small number of pathways (PI3K/Akt, NF-κB, TGF-β/SMAD, VEGF, Wnt/β-catenin) that, taken together, account for most of the regenerative biology described in the literature.
Figure 2. Six of the most-studied miRNAs in MSC exosomes, with their direct targets, signaling pathways, biological effects, and clinical relevance.

miR-21: Anti-Apoptotic and Anti-Fibrotic
miR-21 is one of the most abundant miRNAs in MSC-derived exosomes and is upregulated in human umbilical cord MSC exosomes alongside miR-146a and miR-181 [6]. Its primary targets are PTEN and PDCD4, both negative regulators of the PI3K/Akt pathway. By suppressing them, miR-21 promotes cell survival, proliferation, and migration. In a 2022 study, human umbilical cord MSC small EVs accelerated corneal epithelial wound healing through delivery of miR-21 and activation of the PTEN/PI3K/Akt axis [7]. miR-21 also targets TGFBR2: in a 2021 Stem Cell Research and Therapy study, UCB-MSC exosomes carrying miR-21-5p and miR-125b-5p inhibited TGF-β receptor signaling, suppressed myofibroblast differentiation, and reduced scar formation in wound healing models [8]. The clinical relevance is concentrated in keratinocyte proliferation, anti-fibrotic remodeling, and wound healing applications.
miR-146a: NF-κB Attenuation and M2 Polarization
miR-146a targets IRAK1 and TRAF6, two adapter proteins in the NF-κB inflammatory signaling cascade. By suppressing them, miR-146a attenuates pro-inflammatory cytokine production and shifts macrophages toward the anti-inflammatory M2 phenotype [9]. In MSC exosomes, miR-146a is one of the miRNAs responsible for the conversion of macrophages from M1 to M2, alongside miR-9, miR-125b, miR-127, and miR-155 [9]. A 2021 study in Frontiers in Cell and Developmental Biology demonstrated that hydrogen-peroxide-induced senescence in MSCs reduces exosomal miR-146a content, and that this loss substantially impairs the pro-angiogenic, pro-wound-healing capacity of the resulting exosomes [10]. Knocking down miR-146a in fresh MSC exosomes reduced their angiogenic capacity, while overexpressing it in senescent MSC exosomes partially rescued the impairment [10]. Engineered miR-146a-loaded exosomes have shown pronounced anti-inflammatory and regenerative effects in diabetic wound healing models [11]. miR-146a is the cargo to look for in any product positioned for inflammation, post-procedural recovery, or chronic-wound applications.
miR-126: The Endothelial-Specific Pro-Angiogenic miRNA
miR-126 is the endothelial-specific miRNA most directly responsible for vascular integrity and angiogenesis. The original 2008 work from Wang and colleagues in Developmental Cell identified miR-126 as a regulator of endothelial response to VEGF, demonstrating that miR-126 represses two negative regulators of the VEGF pathway: SPRED1 (a Sprouty-related protein) and PIK3R2 (the p85-β regulatory subunit of PI3K) [12]. Knocking down miR-126 in zebrafish embryos produced loss of vascular integrity and hemorrhage. In MSC exosomes, miR-126 has been shown to accelerate angiogenesis in diabetic rat wounds, and a 2021 study in the Journal of Cellular and Molecular Medicine showed that exosomes from miR-126-overexpressing bone marrow MSCs promoted HUVEC proliferation, migration, and tube formation through PIK3R2-mediated PI3K/Akt activation [13]. A separate study showed that MSC-derived exosomes ameliorate hypoxia/reoxygenation-injured endothelial cells by transferring miR-126 through PI3K/Akt/eNOS signaling [14]. For aesthetic medicine practitioners, miR-126 is the cargo most directly implicated in the post-laser, post-microneedling, and post-procedural revascularization story.
miR-125b and miR-181: Anti-Scar and Anti-Inflammatory Modulators
miR-125b targets TGFBR1 and, alongside miR-21, contributes to the TGF-β/SMAD axis suppression that produces a scar-resistant healing phenotype [8]. miR-181 dampens TLR4 signaling and reduces neutrophil and macrophage recruitment in early injury, complementing miR-146a's later-phase polarization effect [6]. Both miRNAs are upregulated in human umbilical cord MSC exosomes and contribute to the broader anti-inflammatory and remodeling profile that distinguishes UC-MSC products in the literature.
miR-218-5p: The Hair-Restoration miRNA
Hair follicle dermal papilla biology operates through Wnt/β-catenin signaling, and miR-218-5p sits squarely in that pathway. A 2020 study in Science Advances demonstrated that miR-218-5p in dermal papilla cell exosomes targets SFRP2, a Wnt signaling inhibitor, and that miR-218-5p mimics promote hair follicle development while inhibitors block the telogen-to-anagen transition [15]. A 2025 systematic review in trichology consolidates the broader picture: MSC, ASC, and DPC-derived exosomes consistently activate Wnt/β-catenin, VEGF, and PI3K/AKT pathways and drive the transition from telogen to anagen, with promising preliminary clinical data in androgenetic alopecia, alopecia areata, and chemotherapy-induced alopecia [16]. miR-218-5p is the cargo most tightly tied to hair restoration outcomes.
Practitioner takeaway: If a supplier cannot tell you, on the COA or on request, what tissue source produced the exosomes and what the dominant miRNA profile is in their preparation, you do not have enough information to choose the right product for your application. Cargo varies by tissue source, donor, passage number, and culture conditions.
CELL SOURCE
Source Matters: Bone Marrow, Adipose Tissue, and Umbilical Cord
MSC-derived EVs account for approximately 61% of interventional EV studies registered on ClinicalTrials.gov, but "MSC" hides a meaningful biological distinction. A 2020 proteomic study in Stem Cell Research and Therapy identified 1,014 proteins across exosomes derived from human bone marrow, adipose tissue, and umbilical cord MSCs and found that, despite shared functions in extracellular matrix receptor pathways, each source had a distinct functional emphasis [17].
Figure 3. Functional differentiation across the three dominant MSC sources used in clinical exosome production. Source determines cargo, and cargo determines biology.
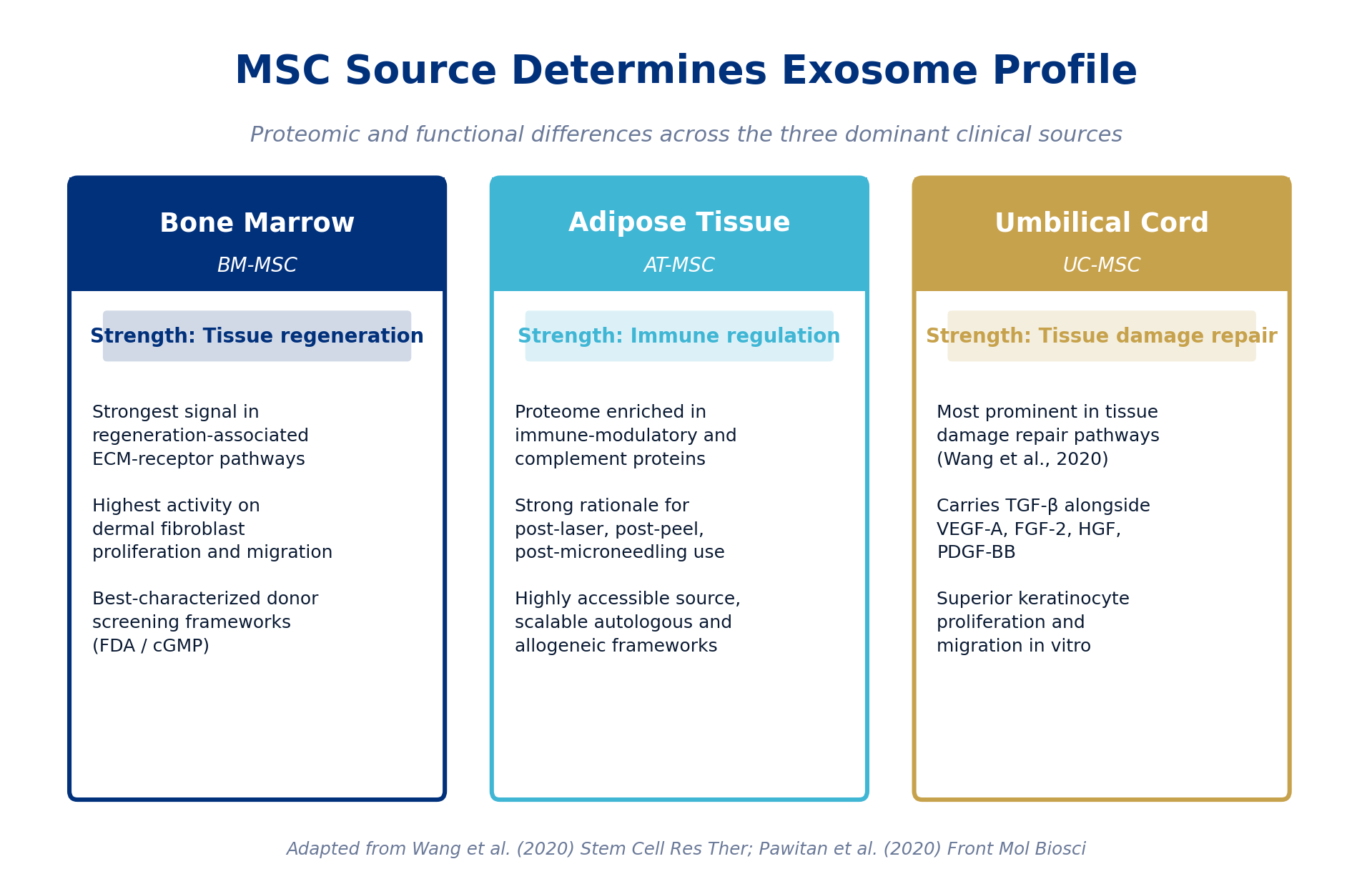
Bone marrow MSC exosomes showed the strongest signal in regeneration-associated pathways, and a 2020 wound-healing study in Frontiers in Molecular Biosciences confirmed BM-MSC exosomes had the highest activity on dermal fibroblast proliferation and migration under serum-free conditions [18]. Adipose tissue MSC exosomes were enriched for immune-modulatory and complement-pathway proteins [17], which is the biological profile that fits aesthetic-medicine post-procedural recovery use cases such as post-laser, post-peel, and post-microneedling. Umbilical cord MSC exosomes were the most prominent in tissue damage repair pathways [17] and were the only source carrying TGF-β alongside VEGF-A, FGF-2, HGF, and PDGF-BB; they also showed superior keratinocyte proliferation and migration in vitro [18].
A 2021 proteomic comparison in the Iranian Biomedical Journal between UC-MSC and AD-MSC exosomes identified 198 differentially expressed proteins and confirmed that UC-MSC exosomes are more strongly enriched in immune, complement activation, and protein activation cascade pathways, while shared functions concentrate around platelet activation and coagulation cascades [19]. For practitioners, the practical implication is straightforward: a generic "MSC exosome" claim is not sufficient. Tissue source, donor selection, and culture conditions are first-order variables that affect what a clinic is actually buying.
CLINICAL EVIDENCE
What the Trials Are Showing in Aesthetic and Dermatologic Applications
The clinical trial landscape for exosomes in aesthetic medicine and dermatology is real, growing, and mostly at Phase 1 and Phase 2. A 2024 review in Skin Health and Disease identified four prospective clinical trials in skin rejuvenation. Park and colleagues conducted a 28-subject prospective, randomized, split-face study showing that human adipose stem cell exosome solution combined with microneedling significantly outperformed microneedling alone on the Global Aesthetic Improvement Scale at 6 weeks, with measured improvements in collagen content, wrinkle reduction, elasticity, hydration, and dyspigmentation through 12 weeks [20]. Proffer and colleagues conducted a 56-participant trial of topically applied human platelet-derived exosome serum and reported significant improvements in erythema, pigmentation, and wrinkle scoring over 6 weeks [20].
A 2025 split-face investigator-blinded non-inferiority trial published in the Journal of Cosmetic Dermatology compared adipose MSC-derived exosomes against platelet-rich plasma in radiofrequency microneedling treatment for photoaged facial skin, the first head-to-head comparison of these two regenerative modalities in aesthetic dermatology [21]. A 2025 systematic review of EV clinical trials in skin aging and regeneration in the International Journal of Molecular Sciences synthesized human and high-translation preclinical data through November 2024, characterizing the platelet-derived exosome category as showing consistent, reproducible aesthetic outcomes across multiple studies [22].
On the hair restoration side, a 2025 systematic review in the World Journal of Stem Cells consolidated the case for stem cell-derived exosomes in androgenetic alopecia, with hair density and thickness improvements supported by Wnt/β-catenin pathway activation and a favorable safety profile across small-cohort clinical reports [23]. The 2024 review in The Aesthetic Surgery Journal positioned topical exosomes alongside topical peptides as the most actively developed acellular biologic platforms in dermatology, with neither yet FDA-approved but with measurable preliminary outcomes across multiple endpoints [24].
The evidence base is genuine, the mechanism of action is well-supported by peer-reviewed biology, and the early clinical signal is consistent. Large-scale Phase 3 placebo-controlled trials remain the next benchmark.
QUALITY DOCUMENTATION
Reading the COA: Twelve Parameters, In Order
A Certificate of Analysis is a structured document. Reading it correctly means knowing what category of information sits where, what units are appropriate for each measurement, and what method was used to obtain the result. The framework that follows aligns with MISEV 2023 [5] and with standard FDA biologic release specifications [25]. A research-grade or clinical-grade COA will address each of these parameters with method, result, and lot specificity.
Figure 4. The twelve parameters every chemist asks before approving an exosome supplier. Failure on any one of these parameters should prompt a follow-up to the supplier before the product enters a clinical workflow.

01 Cell Source Disclosure
The COA should disclose the producing cell type (MSC, platelet, dermal papilla, plant, or other), the tissue of origin (bone marrow, adipose, umbilical cord Wharton's jelly, or peripheral blood), the donor screening criteria applied, the passage number at which exosomes were harvested, and the culture media composition. Xeno-free culture conditions are preferred for clinical-grade products. Source variables affect cargo composition directly, as the proteomic and miRNA literature establishes [17, 18].
02 and 03 Particle Concentration and Size Distribution by NTA
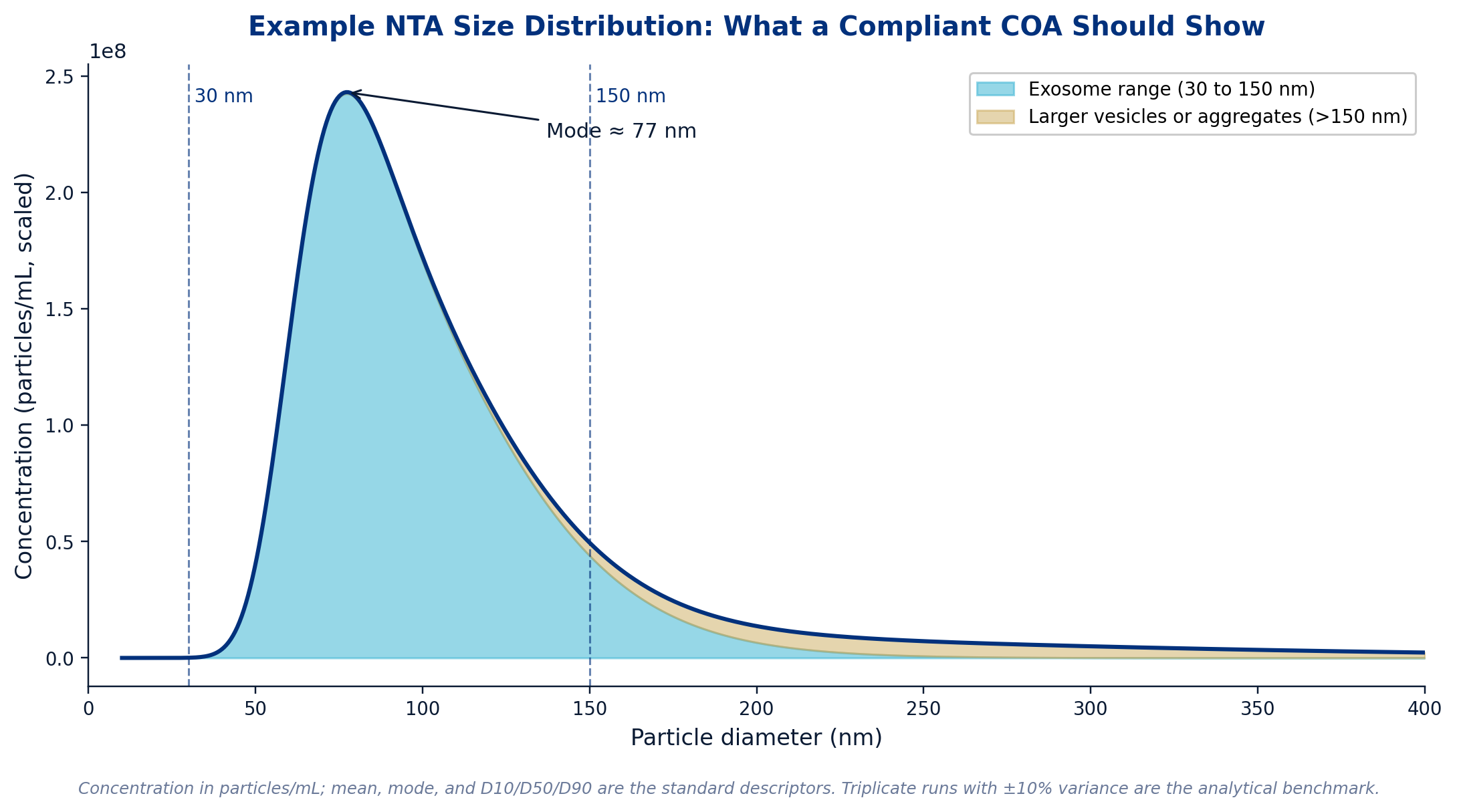
Nanoparticle tracking analysis (NTA) is the standard method for both particle concentration and size distribution. The COA should report concentration in particles per mL, with a working range that places the sample within the validated limits of the instrument (typically 10⁶ to 10¹⁰ particles per mL for a Zetaview or NanoSight class instrument). The size distribution should include mean, mode, and ideally the D10, D50, and D90 percentiles, with the bulk of the distribution falling within the exosome range of 30 to 150 nm. A reputable supplier will provide the raw NTA data, video files, or histogram, not just a single number. Triplicate runs with ±10% variance across replicates is the analytical benchmark for batch-to-batch comparability.
Figure 5. An example NTA size distribution from a compliant exosome preparation. The peak sits within the 30 to 150 nm exosome range, with a tail of larger vesicles and aggregates that should be quantified rather than ignored.
04 Positive Surface Markers
The COA should report at least three positive markers from the MISEV 2023 panel: CD9, CD63, and CD81 are the canonical triad, optionally supplemented with TSG101 or ALIX [5]. Detection should be by Western blot, flow cytometry, or fluorescent NTA. A single tetraspanin reported in isolation does not meet the MISEV minimum and should prompt a request for the missing markers. Surface marker results should be qualitative (positive or negative) at minimum, with quantitative percentage-positive results from flow cytometry preferred when available.
05 Negative Markers
MISEV 2023 requires at least one negative marker to confirm low cellular debris contamination. Calnexin, an endoplasmic reticulum chaperone, and GM130, a cis-Golgi marker, are the most commonly reported negative controls [26]. For blood-derived preparations, apolipoproteins A1, A2, and B, plus albumin, are appropriate negative markers since their presence would indicate plasma protein contamination [26]. The combined presence of ALIX, CD81, and CD9 together with the absence of albumin and calnexin confirms endosomal origin and high purity [26].
06 Sterility (USP <71>)
For any product intended for use in or near a treatment room, sterility testing should follow USP <71>, the standard 14-day aerobic and anaerobic growth assay [27]. The COA must report "no growth" and the result must be lot-specific, not a general claim that the product line has been "sterility tested" historically. Suppliers operating under 503B outsourcing facility registration carry the most rigorous sterility framework in the United States compounding landscape.
07 Endotoxin (USP <85> or USP <86>)
Bacterial endotoxin testing is mandatory for any injectable or parenteral biologic, and is also strongly recommended for topical preparations applied to broken or compromised skin (post-microneedling, post-laser, post-peel). The standard methods are the Limulus Amebocyte Lysate (LAL) assay per USP <85> or the recombinant Factor C (rFC) or recombinant Cascade Reagent (rCR) methods per USP <86> [28, 29]. Results should be quantitative (in EU/mL), with a route-appropriate limit. The conventional limit for parenterals is below 5 EU/kg/hr, and the FDA accepts rFC as an equivalent alternative when method validation has demonstrated equivalence [28].
08 Mycoplasma
Cell-culture-derived biologics carry a real risk of mycoplasma contamination from the producer cells. Routine mycoplasma testing per EP 2.6.7 (PCR or culture-based) must be in the manufacturing release framework [27]. The COA should report a negative result, lot-specific.
09 and 10 Residual Host DNA and Protein Content
Residual host DNA should be quantified, ideally below 10 ng per dose for biologic products as a standard FDA expectation. Total protein content (typically by BCA assay) provides the denominator for a critical purity metric: the particle-to-protein ratio. A high particle-to-protein ratio suggests a clean preparation; a low ratio suggests significant non-vesicular protein contamination from culture media or cell lysate.
11 Storage and Stability
Exosome preparations should be frozen-stored, ideally at minus 80 degrees Celsius, and the COA or accompanying supplier documentation should include stability data: the number of months the product has been validated as biologically active under the specified storage conditions. Cold-chain validation extends to shipping documentation, with temperature loggers preferred for high-value clinical lots.
12 Manufacturing Facility Registration
FDA-registered manufacturing facilities are verifiable through the FDA's establishment registration database. For sterile injectable preparations in the United States, 503B outsourcing facility registration represents the current regulatory benchmark. ExaVeyra has covered the practical distinction between 503A and 503B compounding pharmacies in detail elsewhere on the ExaBlog. Practitioners sourcing exosome products for clinical use should confirm the manufacturing facility's registration status before approving any new supplier.
APPLICATION MATRIX
Distinguishing Use Cases by Practice Type
The right exosome product depends on the application, and the same COA framework applies across practice types with different emphasis. A med spa selecting a topical post-microneedling serum has different priorities from a dermatology office running a hair restoration protocol, which has different priorities from a clinical trial team running a Phase 2 study. The biology and the documentation requirements scale with the application.
Figure 6. The appropriate exosome profile by clinical setting. In the United States, exosome products for human use are FDA-regulated, and topical use carries a different risk profile than injection.

Med Spa: Topical, Post-Procedural, Aesthetic Endpoints
Med spas using exosomes in a topical post-procedural workflow (post-microneedling, post-laser, post-peel, anti-aging serum application) typically benefit from AT-MSC or UC-MSC sourced products with strong miR-146a, miR-21, and miR-126 signal. The relevant outcome measures are erythema reduction, hydration, luminosity, fine line improvement, and patient-reported satisfaction. Topical application avoids the injection-route regulatory profile entirely and aligns with the dominant published clinical trial evidence base [20, 21, 22].
Dermatology: Off-Label Adjunct, Hair Restoration, Scar Revision
Dermatologists working off-label with exosomes typically focus on more targeted indications: photoaged skin, atopic dermatitis adjunct (research only), acne scar revision in combination with microneedling, and androgenetic or alopecia areata hair restoration. BM-MSC exosomes are well-characterized for skin applications [17, 18]. For hair restoration, the signal is concentrated in DPC-derived and ASC-derived exosomes that carry miR-218-5p and activate Wnt/β-catenin [15, 16, 23]. Outcome measures include collagen quantification, elasticity, hair density and thickness, scar resolution, and patient satisfaction.
Clinical Trial / Research: IND or IRB Governance, Full Traceability
Investigators running formal clinical research with exosomes should source from cGMP-grade, FDA-registered manufacturing facilities, with IND-supported product where the trial structure requires it. The 2024 review in the Journal of Pharmacology and Pharmacotherapeutics found that 90 registered human clinical trials involving exosomes have been initiated through 2024, with steady year-over-year increases [30]. As of early 2025, the FDA has approved its first IND for an EV-based candidate [1], and Korea's Ministry of Food and Drug Safety has approved a Phase 1b trial for a cord MSC-derived EV product. For trials, the COA framework above is the floor; investigator brochures, batch retention samples, and full chain-of-custody documentation are the additional layers.
REGULATORY CONTEXT
FDA, MISEV 2023, and the Documentation Floor
In the United States, exosome products intended for human use are regulated by the FDA. Depending on manufacturing method, composition, and intended use, they may be classified under Section 351 of the Public Health Service Act as biologics requiring a Biologics License Application, under 21 CFR Part 1271 as human cells, tissues, and cellular and tissue-based products if they meet the criteria for minimal manipulation and homologous use, or as drug products requiring NDA or IND review. The FDA has issued warning letters to companies making unapproved therapeutic claims for exosome products and has signaled increased enforcement attention to this category.
MISEV 2023 represents the international scientific community's consensus on minimum reporting requirements for studies of extracellular vesicles, and it provides the analytical floor for any commercial COA that claims to characterize exosomes [5, 31]. A supplier whose documentation does not address the MISEV 2023 framework is, in effect, asking practitioners to take vesicle identity on faith. That is the wrong starting position for a clinical product.
Bottom line: The COA is the supplier's quality story in writing. A clinic that knows what to read on it has a defensible procurement decision. A clinic that does not has a marketing document. The chemistry and the regulatory expectations are clear enough that a clinical practice can hold suppliers to a real standard.
EDUCATIONAL DISCLAIMER
This article is intended for licensed medical professionals and is for informational and educational purposes only. It does not constitute medical advice, clinical protocol guidance, or product endorsement. All citations reference peer-reviewed literature available through PubMed or publisher websites. Regulatory status of exosome products is subject to change; consult current FDA guidance and qualified legal counsel.
PEER-REVIEWED REFERENCES
References
[1] Tan F, et al. Clinical applications of stem cell-derived exosomes. Signal Transduction and Targeted Therapy. 2024;9:17. https://www.nature.com/articles/s41392-023-01704-0
[2] Vondrová L, et al. Tetraspanins, More than Markers of Extracellular Vesicles in Reproduction. International Journal of Molecular Sciences. 2020. https://pmc.ncbi.nlm.nih.gov/articles/PMC7589920/
[3] Mathieu M, et al. Differential proteomics argues against a general role for CD9, CD81 or CD63 in the sorting of proteins into extracellular vesicles. Journal of Extracellular Vesicles. 2023. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC10390663/
[4] Ivanusic D, Denner J. The large extracellular loop is important for recruiting CD63 to exosomes. microPublication Biology. 2023. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC10432940/
[5] Welsh JA, et al. Minimal Information for Studies of Extracellular Vesicles (MISEV2023). Journal of Extracellular Vesicles. 2024. https://onlinelibrary.wiley.com/journal/20013078
[6] Fang S, et al. Mesenchymal stem cells-derived exosomal microRNAs contribute to wound inflammation. Science China Life Sciences. 2016. https://link.springer.com/article/10.1007/s11427-016-0240-4
[7] Wang S, et al. Umbilical Cord Mesenchymal Stem Cell-Derived Small Extracellular Vesicles Deliver miR-21 to Promote Corneal Epithelial Wound Healing through PTEN/PI3K/Akt Pathway. Stem Cells International. 2022. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9303509/
[8] Hu Y, et al. Exosomes derived from human umbilical cord blood mesenchymal stem cells stimulate regenerative wound healing via TGF-beta receptor inhibition. Stem Cell Research and Therapy. 2021. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8336384/
[9] He X, et al. Application of mesenchymal stem cell exosomes in the treatment of skin wounds. Burns and Trauma. 2023. https://www.sciencedirect.com/science/article/am/pii/S2590183423000157
[10] Yu Y, et al. Hydrogen Peroxide-Induced Senescence Reduces the Wound Healing-Promoting Effects of Mesenchymal Stem Cell-Derived Exosomes Partially via miR-146a. Aging and Disease. 2021. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7801275/
[11] Wang Y, et al. MiR146a-loaded engineered exosomes released from silk fibroin patch promote diabetic wound healing by targeting IRAK1. Signal Transduction and Targeted Therapy. 2023. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9922687/
[12] Wang S, et al. miR-126 Regulates Angiogenic Signaling and Vascular Integrity. Developmental Cell. 2008;15:261-271. https://www.sciencedirect.com/science/article/pii/S1534580708002876
[13] Zhang L, et al. Exosomes from microRNA-126 overexpressing mesenchymal stem cells promote angiogenesis by targeting the PIK3R2-mediated PI3K/Akt signalling pathway. Journal of Cellular and Molecular Medicine. 2021. https://onlinelibrary.wiley.com/doi/10.1111/jcmm.16192
[14] Pan Q, et al. Exosomes Derived from Mesenchymal Stem Cells Ameliorate Hypoxia/Reoxygenation-Injured ECs via Transferring MicroRNA-126. Stem Cells International. 2019. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6589209/
[15] Hu S, et al. Dermal exosomes containing miR-218-5p promote hair regeneration by regulating beta-catenin signaling. Science Advances. 2020;6:eaba1685. https://www.science.org/doi/10.1126/sciadv.aba1685
[16] Hassanshahi A, et al. Exosomes in trichology: A literature review. World Journal of Stem Cells. 2025. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC12814429/
[17] Wang ZG, et al. Comprehensive proteomic analysis of exosomes derived from human bone marrow, adipose tissue, and umbilical cord mesenchymal stem cells. Stem Cell Research and Therapy. 2020;11:511. https://pmc.ncbi.nlm.nih.gov/articles/PMC7694919/
[18] Pawitan JA, et al. Differential Wound Healing Capacity of Mesenchymal Stem Cell-Derived Exosomes Originated From Bone Marrow, Adipose Tissue and Umbilical Cord Under Serum and Xeno-Free Condition. Frontiers in Molecular Biosciences. 2020;7:119. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7327117/
[19] Bai S, et al. Proteomics Analyses Reveal Functional Differences between Exosomes of MSCs Derived from The Umbilical Cord and Those Derived from The Adipose Tissue. Iranian Biomedical Journal. 2021. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7944129/
[20] Bai S, et al. Clinical applications of exosomes in cosmetic dermatology. Skin Health and Disease. 2024;4:e348. https://onlinelibrary.wiley.com/doi/full/10.1002/ski2.348
[21] Estupiñan B, Ly K, Goldberg DJ. Adipose Mesenchymal Stem Cell-Derived Exosomes Versus Platelet-Rich Plasma Treatment for Photoaged Facial Skin: An Investigator-Blinded, Split-Face, Non-Inferiority Trial. Journal of Cosmetic Dermatology. 2025. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC12104007/
[22] Effectiveness of Extracellular Vesicle Application in Skin Aging Treatment and Regeneration: Do We Have Enough Evidence from Clinical Trials? International Journal of Molecular Sciences. 2025;26:2354. https://www.mdpi.com/1422-0067/26/5/2354
[23] Therapeutic potential of stem cell-derived exosomes in hair regeneration: A systematic review. World Journal of Stem Cells. 2025. https://pmc.ncbi.nlm.nih.gov/articles/PMC12305264/
[24] The Innovative and Evolving Landscape of Topical Exosome and Peptide Therapies: A Systematic Review of the Available Literature. Aesthetic Surgery Journal Open Forum. 2024. https://pmc.ncbi.nlm.nih.gov/articles/PMC11023079/
[25] United States Pharmacopeia. USP <71> Sterility Tests. United States Pharmacopeial Convention. https://www.uspnf.com/
[26] Theofilopoulos S, et al. Exosomal Protein Markers as Potential Non-Invasive Biomarkers for Colorectal Cancer. International Journal of Molecular Sciences. 2025. https://www.mdpi.com/1422-0067/26/22/11060
[27] The Critical Role of Quality Control in Exosome-Based Therapeutics: Standards under USP <71>, USP <85>, and ICH Q5A. https://www.atlantisbioscience.com/blog/quality-control-exosome-therapeutics-academic-innovators/
[28] United States Pharmacopeia. USP <85> Bacterial Endotoxins Test. United States Pharmacopeial Convention. https://www.uspnf.com/
[29] United States Pharmacopeia. USP <86> Bacterial Endotoxins Test using Recombinant Reagents. United States Pharmacopeial Convention. https://www.uspnf.com/
[30] Mohan S, et al. Clinical Frontiers of Exosome Research: A Comprehensive Analysis of Human Trials. Journal of Pharmacology and Pharmacotherapeutics. 2025. https://journals.sagepub.com/doi/10.1177/0976500X251361201
[31] Witwer KW, et al. Minimum information for studies of extracellular vesicles 2023: relevance to cell and gene therapies. Cytotherapy. 2024. https://www.isct-cytotherapy.org/article/S1465-3249(24)00727-8/abstract